 It must be emphasized that measurement does not mean only a process in which a physicist-observer takes part, but rather any interaction between classical and quantum objects regardless of any observer. Indeed the uncertainty principle has its roots in how we apply calculus to write the basic equations of mechanics. Thus, the uncertainty principle actually states a fundamental property of quantum systems and is not a statement about the observational success of current technology. It has since become clearer, however, that the uncertainty principle is inherent in the properties of all wave-like systems, and that it arises in quantum mechanics simply due to the matter wave nature of all quantum objects. Heisenberg utilized such an observer effect at the quantum level (see below) as a physical "explanation" of quantum uncertainty. Historically, the uncertainty principle has been confused with a related effect in physics, called the observer effect, which notes that measurements of certain systems cannot be made without affecting the system, that is, without changing something in a system. The formal inequality relating the standard deviation of position σ x and the standard deviation of momentum σ p was derived by Earle Hesse Kennard later that year and by Hermann Weyl in 1928: 
In the published 1927 paper, Heisenberg originally concluded that the uncertainty principle was Δ p Δ q ≈ h using the full Planck constant. Introduced first in 1927 by the German physicist Werner Heisenberg, the uncertainty principle states that the more precisely the position of some particle is determined, the less precisely its momentum can be predicted from initial conditions, and vice versa. The uncertainty principle implies that it is in general not possible to predict the value of a quantity with arbitrary certainty, even if all initial conditions are specified. 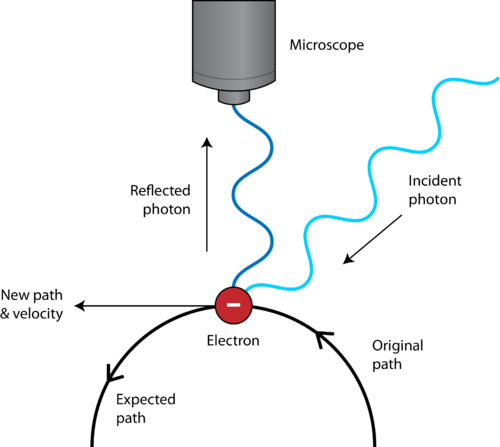
Such variable pairs are known as complementary variables or canonically conjugate variables and, depending on interpretation, the uncertainty principle limits to what extent such conjugate properties maintain their approximate meaning, as the mathematical framework of quantum physics does not support the notion of simultaneously well-defined conjugate properties expressed by a single value. Uncertainty principle of Heisenberg, 1927. Critical reactions The ideal of the detached observer Einstein's slit Einstein's box EPR paradox for entangled particles Popper's criticism Many-worlds uncertainty Free will ThermodynamicsĬanonical commutation rule for position q and momentum p variables of a particle, 1927.History Terminology and translation Heisenberg's microscope.Harmonic analysis Signal processing Discrete Fourier transform Benedicks's theorem Hardy's uncertainty principle.Uncertainty relation with three angular momentum components.Additional uncertainty relations Systematic and statistical errors Quantum entropic uncertainty principle.Examples Quantum harmonic oscillator stationary states Quantum harmonic oscillators with Gaussian initial condition Coherent states Particle in a box Constant momentum. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
